Author: Zoltan Simandi, Ph.D.
Manual vs automated cell counting
The definition of automation is “the creation and application of technologies to produce and deliver goods and services with minimal human intervention”. The motivation behind automating processes is to free employees from tedious work and allow them to focus on more uniquely human tasks. Automation has already revolutionized the operation of research labs in many ways. The polymerase chain reaction (PCR), a laboratory technique that was invented by Kary Mullis in 19851, today is a standard process in every lab. In the beginning, all of the necessary PCR steps had to be performed manually - moving the DNA sample back and forth between large water baths of different temperatures. Thermocycling machines automated this heating and cooling process, making PCR a simple and reproducible process all over the world.
In recent years, cell counting went through a similar development: automated cell counters became very popular, yet many labs are still hesitating to invest in an automated cell counter. The reason is simple – manual cell counting seems to be working “well enough” at the moment for most. Should we continue to perform cell counting manually? Would that prevent us from being more efficient and productive? To decide whether it is time to invest in automated cell counting, we have to consider the following:
- Accuracy: would the automated version produce similar or even more accurate cell counts than traditional manual cell counting?
- Versatility: does automated cell counting have the ability to be adapted to many different applications?
- Reporting: can we improve the way cell counting results are documented?
- Costs/Affordability: what is the maintenance cost?
Only if all these important criteria are met, will we reach the point that automation frees up time and enhances productivity.
-----------------------------------------
With the CytoSMART Exact FL, researchers can apply automated fluorescence cell counting to their laboratory practice. This automated cell counter is equipped with an advanced optical system and highly trained pattern recognition software that generate accurate and reliable cell counts in a fraction of the time. Learn more about it here.
-----------------------------------------
Accuracy
Hemocytometer is the gold standard, most widely used device for visually counting the number of cells. Using a brightfield microscope to visualize a cell population, researchers count cells within defined areas marked on a hemocytometer slide. However, hemocytometer-based cell counts often show low levels of reproducibility and have a number of additional inherent limitations. Firstly, the generated results are subjective and inter-operator variation can be high. The resulting cell counts may differ from person to person, even if they count the very same grid. Secondly, the tendency to count too few cells means that standard errors can be high, and results will lack statistical robustness. Thirdly, hemocytometer-based cell counting typically relies on the use of Trypan Blue, a cell exclusion dye which only stains dead cells. But this dye can be incorporated by live cells as well after a short exposure time2.
One of the most important expectations for automation is to minimize human interventions and eliminate bias and error. However, many users of prior generations of automated cell counters reported that those machines were largely inaccurate, slow, and expensive to operate3. Progressive improvement of these instruments has allowed great precision and speed at a very low cost, and the new generation of automated cell counters has the advantage of largely removing human subjectivity from the cell counting workflow4.
Versatility
A general concept is that manual cell counting is easily adaptable to many situations, making it highly versatile. In contrast, automated cell counting allows for a higher throughput of samples but limits its versatility available with a specific system. In fact, there are many applications where automated cell counters can outperform manual cell counting. For instance, if you need selective cell counting within a size range in a mixed population, it is not achievable by manual cell counting. Thus, the ability to gate for certain cell sizes significantly expands the utility of automated cell counters in certain applications. Another example when automated cell counters can be useful is when we want to obtain accurate cell counts from samples containing clumpy cells or to accurately count spheroids. Some newer automated cell counter models feature pattern-recognition software with de-clustering algorithms that allow to achieve an accurate cell count even with clumpy cells. Samples that contain debris add further complexity to cell counting and the already mentioned gating for a certain cell size range can be really handy in such cases. Automated cell counters have also improved the way to assess the live-dead cell ratio in the sample. Newer models of automated cell counters are equipped with fluorescence optics. This enhancement allows us to quantify multiple labels within the same sample and extract more accurate cell count information.
Overall, it seems that the limited versatility of automated cell counters is no longer true and there is an increasing number of applications when automated counters became the preferred way over manual counting.
Reporting
For manual counting, reporting has not changed since the first hemocytometer was used in the 19th century. At best, the operator simply writes results in their lab notes. On the automated side, a lot has changed, allowing for more in-depth and faster reporting of the cell counts. With current setups, there are more detailed analysis reports available, including generation of a cell size histogram based on (1) cell size; (2) live, dead, and total cell counts; (3) the live and total cell concentration; (4) the mean cell diameter; and (5) the percent viability, just to name a few. Furthermore, with cloud data storage, the operator can access and review the data at any time. This may reveal details on cell morphology or size distribution that otherwise would be easily overlooked at the time of cell counting.
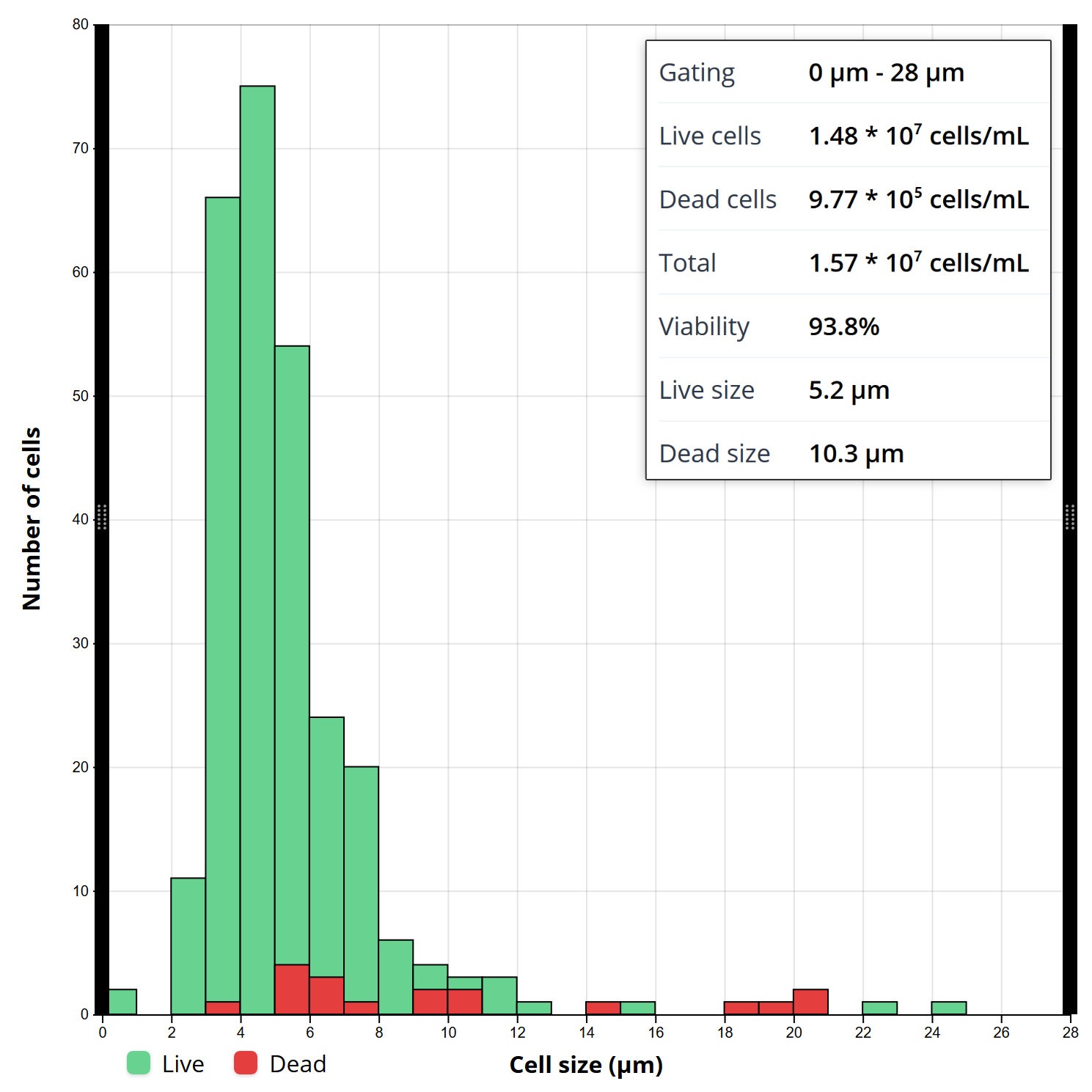
Costs/Affordability
Hemocytometer has a low initial purchase price compared to an automated cell counter, and this makes manual counting attractive to many researchers on a limited budget. However, we must also consider the ongoing costs of the additional labor required and the ultimate price of inaccurate results. Automated cell counters can produce accurate cell counts in less than 30 seconds! Accordingly, the relative cost of automated cell counting greatly decreases in total as the amount of counts increases. One of the biggest additional costs of automated cell counters was the necessity of consumables, such as disposable plastic hemocytometers. These consumables could surpass the initial purchase cost greatly when the device was used for a large number of cell counts. The reusable slides made automated cell counting more cost-effective, and automated cell counting is no longer considered expensive.
Conclusions
Since the operator directly observes the cells, manual counting still has a few important strengths, such as early problem detection, allowing the user to capture any errors quickly. Nevertheless, with all the new features, improved technology and software, automated cell counters have become an essential technology for many researchers. Compatibility of automated cell counters has been verified for use with several commonly used cell lines and primary cell types. Focusing on details of both manual and automated systems regarding their costs, accuracy, versatility, and reporting and how these are related to your requirements will help to decide whether it is time to move on and automate cell counting in your lab.
Table 1 | Comparison of manual and automated cell counting approaches.
| Manual cell counting | CytoSmart Exact FL | |
|---|---|---|
 |
 |
|
| Accuracy | User-to-user variability Human interpretation Trypan blue is less accurate for live/dead cell quantification Multi-count is time-consuming |
Increased counting volume AI-driven image analysis Dual fluorescence cell counting Option for multi-count |
| Versatility | Easly adaptable to many applications Difficult to differentiate between cells and debris |
Declustering algorithm Module for counting organoids Counts cells down to 4 µm in diameter |
| Reporting | Manual data recording Can not meet with stringent documentation Measurement time: 3-5 minutes |
Connected to the CytoSmart cloud environment Data formats: JPG, CSV, PDF (Pre)dilution calculator Measurement time: 3 sec |
| Affordability | Low initial purchase price Inexpensive maintenance |
Compatibility with reussable counting chambers makes it inexpensive Works with any cell counting chamber (0depth of 0.1 mm) |
| Recommended for those who only need to count occasionally | Great for those that require a reproducible, traceable measurement |
A final tip
When trying a new setup, always try to arrange a demonstration. With a trial, you can see if the product fits your needs.
----------
Learn more about the CytoSMART cell and organoid counting solutions here.
