Clinical use, isolation, and cell culture methods
Human umbilical cord mesenchymal stem cells (hUC-MSCs) have been used as a promising stem cell source in clinical trials. A major advantage of using hUC-MSCs is that donor material is continuously available and isolation is not limited by ethical issues. Harvesting is non-invasive and relatively straightforward3,4. hUC-MSCs display superior properties in immunomodulation, differentiation, and self-renewal capacity compared to adult mesenchymal stem cells (MSCs)1,2.
In this article, we will discuss the applications, isolation, and culturing of hUC-MSCs. It will become clear that following a strict culturing protocol, sticking to specific seeding densities, and observing confluence is essential to maintaining stemness of the MSCs.
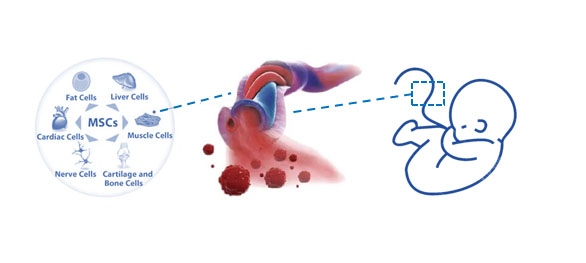
Clinical use of human umbilical cord mesenchymal stem cells
Currently, in April 2020, 241 clinical trials using hUC-MSCs are registered, of which 56 are in the recruiting phase and 46 have been completed (source: clinicaltrials.gov). Cell therapies using hUC-MSCs are being performed for diseases like diabetes7, liver failure8, hemorrhagic stroke9, and orthopedic diseases10, to name a few. We recommend the reviews by Davies et al. 2017, who provide an overview of organs that are targeted in clinical trials, and Ding et al. 2015, who provide a concise description of the general uses and benefits of hUC-MSCs like biobanking, anticancer effects, and immune modulation3,4.
Side note: the umbilical cord blood is a source of hematopoietic stem cells (HSC). In the review by Dessels et al. 2018 the potential and current status of hUC-HSCs in transplantation for regenerative purposes is discussed11.
Next to cell therapy studies, hUC-MSCs are used to obtain stem-cell-derived extracellular vesicles (EVs)12. EVs are comprised of exosomes and microvesicles that are part of the paracrine signaling functions of stem cells. Using EVs as opposed to the cells themselves can provide a better safety profile since these should not display immune-related side effects of cell transplantation13.
So why use hUC-MSCs rather than another source? For any stem cell application, it is imperative to look at the feasibility of cell isolation and expansion.
BM-MSCs are the most commonly studied source of MSCs ever since they were isolated from mice by Friedenstein et al. 1966. While the bone marrow has been a valuable source of MSCs, the characteristics of which have been studied extensively, using hUC-MSCs has several benefits.
To use MSCs for transplantation, large quantities of cells are needed, so ideally donor material contains many MSCs to provide a nice starting point. The cell-fraction of MSCs in the UC lies between 1:333 – 1:160914, which is a high density compared to MSCs from BM, which has an MSC cell fraction of 1:25000 – 1: 10000015. Next to that, using the UC is much more convenient when compared to bone-marrow MSCs (BM-MSCs) harvesting, which is invasive and painful. Additionally, hUC-MSCs retain proliferative capacity, which is exemplified by a steady doubling time for up to ten passages5, whereas BM-MSCs lose stability after six passages16. Moreover, hUC-MSCs are less likely to develop teratomas17, have a larger differentiation potential, and display relatively fast self-renewal rates18. Due to the lack of HLA-DR antigens, the immunomodulation capacity of hUC-MSCs seems more suitable than BM-MSCs19,20. These superior features of hUC-MSCs compared to BM-MSCs demonstrate that hUC-MSCs are an attractive stem cell source.
Several protocols for harvesting and expanding hUC-MSCs are known, these show that MSC stemness remains stable and cell proliferation can be maintained for numerous passages. Studies have been performed where hUC-MSCs have been kept in culture for 194 days5 and up to fifteen passages6, while retaining MSC markers. In the sections below, we will go into the steps for dissecting, isolating, and culturing UC-MSCs.
UC-MSC isolation methods - anatomical sections of the umbilical cord
Isolation of hUC-MSCs is performed by slicing the donor material into 3 cm fragments, removing the blood, and washing for approximately an hour. The fragments are then cut and treated according to their respective isolation protocol (see examples below). The cells are strained, centrifuged, and re-suspended in medium21.
For the initial extraction several anatomical locations within the umbilical cord can be used to obtain MSCs (see Figure 1). Next to the perivascular (PV) region and cord lining (CL), the Wharton's Jelly (WJ) derived MSCs are most commonly used and it has been postulated that WJ-MSCs display superior quality over the other regions in the umbilical cord. In a study by Mennan et al. 2013, it was shown that WJ-MSCs have excellent osteogenic and adipogenic differentiation potential when compared to other anatomical locations of the umbilical cord22,23.
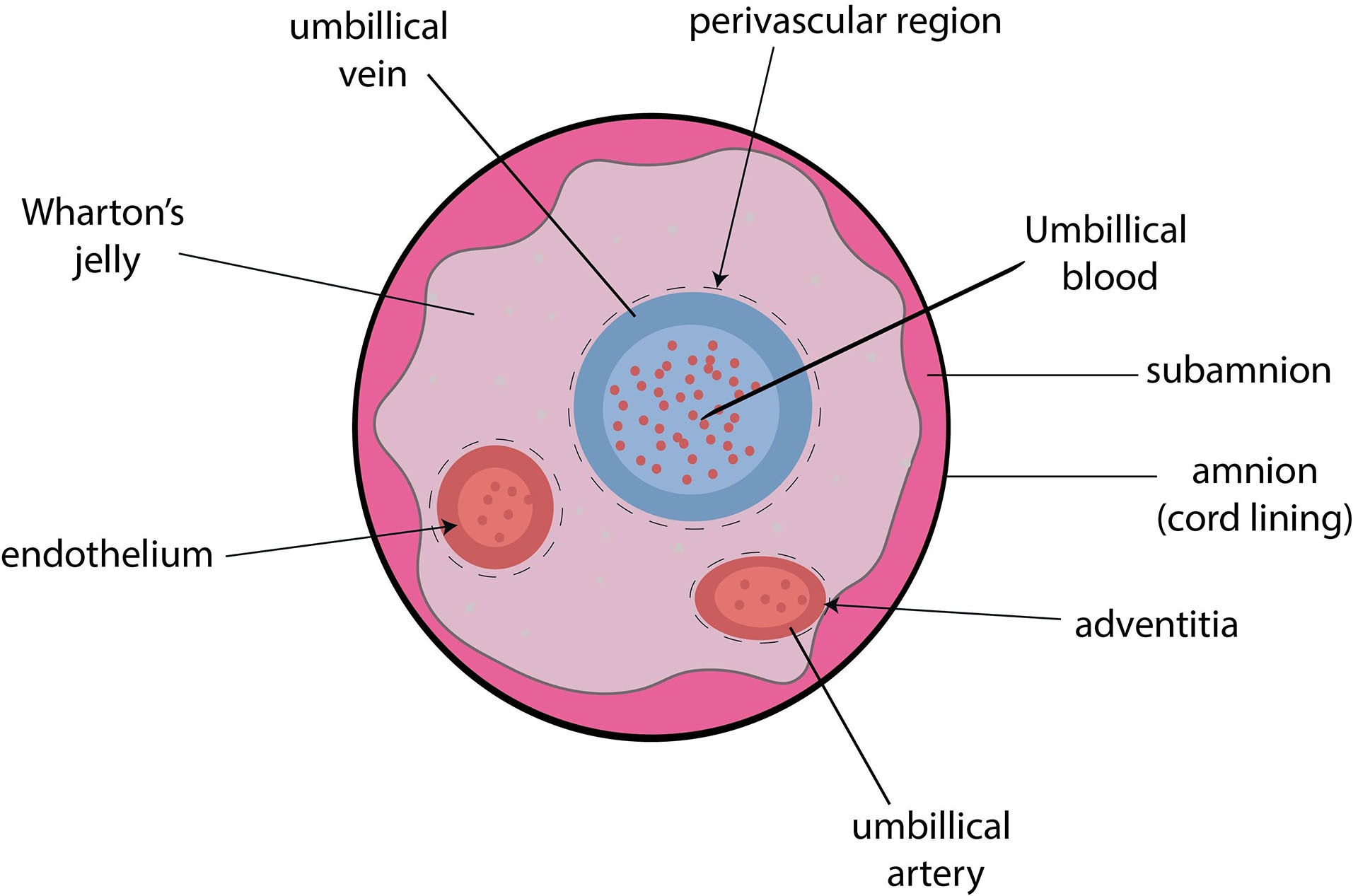
Conversely, in a study by Russo et al. 2020, UC-MSCs from three different anatomical regions were compared to validate their potential to treat ischemia-related injuries. It was hypothesized that there would be a difference in cellular response, related to the anatomical region the MSCs were harvested from. The cells were tested in environments that were poor in nutrients and oxygen. Initially, cells were analyzed for markers that are thought to decrease the immunogenic response after being kept in a hypoxic environment. Downregulation of the immunogenic response is an important factor in containing the tissue damage caused by ischemic injuries. No significant differences between the cell populations were observed. Furthermore, cell metabolism and survival between populations were comparable in both hypoxia and normoxia conditions. The mitochondrial activity and the response to glucose and oxygen deprivation were measured. Interestingly, the PV- and CL-MSCs even outperformed the WJ-MSCs in these experiments24.
----------------------
Also see the CytoSMART article that discusses important parameters of cell culturing when performing cell metabolism studies.
----------------------
To isolate the MSCs from the dissected tissue three methods are commonly used: enzymatic, enzymatic explant, and the explant method. The enzymatic method being the fasters, followed by the enzymatic explant and the explant method being the slowest. Even though the explant method is time-consuming, it delivers stable cells with desired properties. The enzymatic methods are faster but are not as stable8.
- The enzymatic digestion is by far the fastest method of the three, taking approximately 3 hours to complete, with a 45 min step for enzymatic digestion25. Targeting different anatomical areas of the UC is possible26. In some cases only the mid-section of the UC is used, also referred to as the UC parts culture, which has an even shorter digestion time.
- The explant isolation method is slow, but is not prone to biological contamination, as opposed to the enzymatic digestion method, which has multiple handling steps. Small 3-5 mm pieces are kept in culture for 7-10 days after which the MSCs are extracted. This method is straightforward and provides a homogeneous cell population that has good proliferation capacity29.
- The enzymatic-explant isolation method is the middle ground between the two methods mentioned above. Mild treatment with enzymatic digestion loosens the UC tissue and speeds up MSC outgrowth27.
Maintaining UC-MSC stemness in culture
Cryopreservation is a viable option to keep cells stable, which is thought not to hamper plating efficiency or phenotype. In principle the UC-MSCs are seeded and can be maintained for about 10 to 15 passages, depending on the final application.
Some studies have demonstrated that exceptionally long culture periods are possible. In a study by Otte et al. 2013, it was found that that MSC stemness was retained by keeping the UC tissue in the cell culture. MSCs were kept in the initial culture for up to 194 days, where a medium change was done every 5 days. After this period cells could be extracted and passaged ten times before losing the minimal MSC marker requirements28. Otte et al. 2013 postulate that keeping UC tissue in culture provides a micro-environment for the MSCs that helps to retain their stemness5.
When trying to keep the MSCs in a steady proliferative state, making sure that cells proliferate at a slow pace seems to be beneficial. In that sense, we can draw a comparison with the extraction methods, while the explant method is the most time-consuming, it does lead to a higher yield. Let’s take a look at an example study performed by Nekanti et al. 2010. They observed that a seeding density of 1000 cells/cm2 showed steady doubling rates, in contrast to cellular densities from 2000-5000 cells/cm2. Low seeding density samples displayed 4 cumulative population doublings per passage at 1000 cells/cm2 and 3 or fewer in the 2000-5000 cell/cm2 seeding densities. Corresponding to a yield that was a factor two larger in the final passage for the low seeding density cultures2.
Another straightforward way to ensure stemness is to prevent the over-growing of cell cultures. If the cell population becomes quiescent, usually at 90-100% confluence, cells start to lose MSC-specific markers and display limited proliferative capacity.
-------------------------
For researchers that are performing side-by-side comparison studies of cell cultures and want to make time-lapse videos to observe the growth of cell populations, we recommend looking at the CytoSMART Lux2 Duo Kit.
-------------------------
Maintaining stemness by closely following cell growth
Human umbilical cord mesenchymal stem cells have gained popularity in recent years. Many studies have led to a consensus on how to extract, expand and, if needed, to differentiate stem cells. Maintaining stemness requires careful monitoring of culture conditions.
The potential that these cells have in therapeutics makes these efforts worthwhile. Observing steady growth rates throughout the expansion procedure is a viable way to validate the desired quality of hUC-MSCs cultures.
Expanding MSCs from donor tissue up to a few billion cells can easily take a month2. While these cells are relatively stable, preventing differentiation during the expansion process is not straightforward. There is a limit to the number of cell passages before the cells lose their original multipotent properties.
Considering the long culture period and the challenge to retain MSCs stemness, it is important to carefully control the cell culture conditions. If batches lose their desired properties due to slight errors or unforeseen variations in growth speed, this can result in losing weeks of work and render donor material useless. If cell cultures are over-grown to confluence levels of 90-100% cells can (1) go into quiescence, (2) stop proliferating, and (3) differentiate into unwanted cell types (e.g. adipose instead of osteogenic).
We propose an imaging tool (CytoSMART Lux2) that can help to keep track of growth rates of cell cultures containing hUC-MSCs. This way researchers can continuously track and evaluate the proliferation rate of the culture and monitor cell health. Monitoring cell culture growth allows researchers to rationally time the cell expansion. Automated imaging of this process can assist standardization of operating procedures in an unambiguous manner and help documentation of the conditions of cell cultures.
Related Products
There are currently no products tagged to this resource.